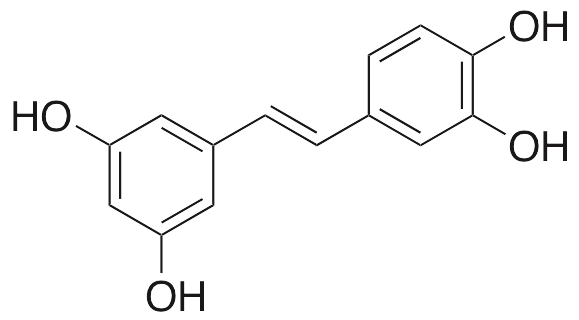
Description
Piceatannol is a polyphenolic hydroxystilbene derivative of resveratrol with increased bioavailability and bioactivity compared to its parent compound. Piceatannol exhibits pro-angiogenic, anticancer, antioxidative, and anti-obesity properties. Piceatannol directly inhibits HIF-prolyl hydroxylase-2 (HPH-2), increasing levels of HIF-1α, VEGF, and heme oxygenase-1 (HO1). When complexed with copper (II), the piceatannol-Cu complex increases ROS levels and damages DNA. Additionally, piceatannol suppresses nuclear translocation of p65 and p50, preventing activation of NF-κB and downregulating expression of matrix metalloproteinase 9 (MMP9). Piceatannol also downregulates phosphorylation of Akt in prostate cancer cells. In other in vitro models, this compound increases levels of tissue inhibitor of metalloproteinase 2 (TIMP2) and inhibits phosphorylation of STAT3; it also downregulates Bcl-xl and inhibits phosphorylation of JNK. In prostate cancer cells, piceatannol decreases levels of cyclins A and D1 and also decreases cyclin-dependent kinase 2 and 4 (CDK2, CDK4) activity, inhibiting cell growth. The hydroxyl groups on piceatannol function as radical scavengers, preventing accumulation of ROS and RNS. This compound noncompetitively inhibits the insulin receptor (IR), preventing phosphorylation of the receptor, IR substrate-1 and Akt, inhibiting adipogenesis of preadipocytes; it may also directly inhibit PI3K and JAK1.