Description
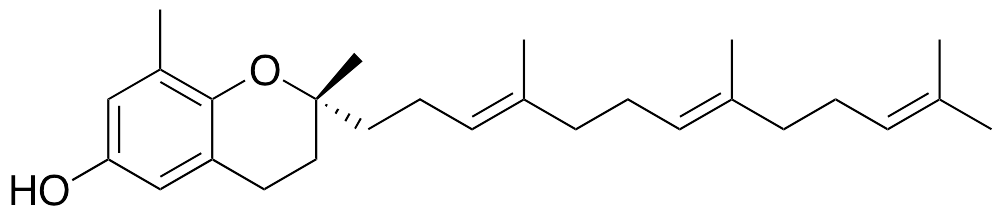
Tocotrienols contain an arene ring, a chromanol ring, and an isoprenoid tail; all tocotrienol compounds contain a methyl group on C8 of the arene ring. δ-Tocotrienol contains only the methyl group at C8; all other available sites are substituted with hydrogen atoms. Tocotrienols are members of the vitamin E family, typically found in vegetable oils, nuts, and grains. Tocotrienols exhibit strong antioxidative activity as well as anti-angiogenic, anticancer, and anti-hyperlipidemic qualities. Of all of the tocotrienols, δ-Tocotrienol displays the strongest anti-angiogenic and anticancer activities. Tocopherols, common substituents of over-the-counter vitamin E supplements, interfere with tocotrienol activity and show weaker antioxidative effects. Tocotrienols exhibit significant radical recycling abilities in cellular models of lipid peroxidation and oxidative damage and suppress HMG-CoA reductase activity. In vitro, these compounds induce apoptosis through inhibition of Id1, EGFR, and NF-κB; they also inhibit vessel formation and proliferation in aortic endothelial cells.